GREAT NEWS! Rapid Antigen testing for COVID-19 now available. Vaccination Could Be Starting Soon…Stand By
Pfizer’s coronavirus vaccine could be approved and ready for distribution by the second week of December. This was announced on various network news programs on Sunday, November 22 by Operation Warp Speed Chief Science Advisor, Moncef Slaoui.
The goal, he said, was to immunize 20 million people by the end of December and vaccinate another 30 million people every month after that.
“We can ship vaccines to the immunization sites within 24 hours from approval,” said Slaoui. Pfizer’s application for emergency use is to be reviewed by the FDA on December 10. It will evaluate the Moderna vaccine on December 17.
About 70% of the country ― or about 230 million people ― would need to take a coronavirus vaccine to achieve “true herd immunity,” he said, which could happen by May 2021.
Slaoui said he felt comfortable with the safety of the vaccines and pledged to make sure the data is transparent. The CDC is issuing guidance to states about which groups should be first to get it, and the states are determining how to distribute the vaccine at the local level. Health care workers and vulnerable groups who face the highest risks for contracting severe COVID-19 will receive a vaccine first.
He expressed concern about the hesitancy to receive the vaccine on the part of the public. “I think it’s very unfortunate because this has been exacerbated by the political context under which we have been working”
==================================================================
Sources:
CNN: “Vaccine czar: ‘No political interference’ with vaccine.”
ABC News: “Operation Warp Speed adviser concerned about vaccine skepticism ‘exacerbated’ by ‘political context.'”
NBC News: “Full Operation Warp Speed Chief: ‘Within 24 hours’ of approval, vaccine will be available to states.”

Although this Thanksgiving is turning out to be a challenging one, I am very grateful that we certainly do have something to be very thankful for. That is the loyalty and patronage of our patients.
Wishing you and yours a very happy and healthy Thanksgiving holiday.
~ Alla Weisz, M.D., M.B.A.
Please pay a virtual visit to our Thanksgiving-decorated offices by clicking the arrow below. Afterwards, hit the BACK arrow to return to this newsletter:

Major Weight Loss Breakthrough Announced

A study measuring the effectiveness of Semaglutide, a new weight loss medication made by Novo Nordisk, has shown outstanding results. “This is a potential major breakthrough,” in obesity management according to the investigators.
Six hundred and eleven overweight (but non-diabetic) adults were studied for a period of 68 weeks. The patients receiving Semaglutide experienced a 10 percentage point greater weight loss than those who received a placebo.
Even more impressive was the finding that a full 75% of the patients lost 10% or more of their starting body weight. 55% lost more than 15% and 36% lost more than 20%. These results were dramatically greater than those observed in the placebo control group. In addition to the medication (or placebo) both groups received regular evidence-based counseling and were placed on a diet regimin.
“The real question, though, is what comes next,” stressed Dr. Scott Kahan, the director of the National Center for Weight and Wellness, Washington, DC.
“Will it be approved by the FDA? I believe it will” he told Medscape Medical News in an email. “Yet we already have several effective obesity medications approved over the past decade — all of which are rarely used and therefore make little impact for patients in the real world.”
“Will there be insurance coverage, and therefore practical access for those who could most benefit?” he continued. “Will prescribers counsel their patients about obesity management, including the use of effective medications? Will patients utilize available options?”
“These and other fundamental issues must be figured out before we anoint any treatment option as a meaningful step forward, let alone a transformative development,” according to Kahan.
Previous studies have shown that intensive behavioral therapy and medication have additive effects. This was demonstrated in this study that involved 611 adults (81% women) who had an average age of 46 and had a mean body mass index (BMI) of 38 kg/m2. A BMI of 30 or more is classified as obese.
All participants received intensive behavior therapy sessions provided by a registered dietitian, which typically lasted 20-30 minutes each and were given weekly for 12 weeks, every other week for the next 12 weeks, and then monthly. The dietitian gave participants behavioral strategies to help them stick to their diet and physical activity goals.
During the first 8 weeks, participants were provided with a 1000-1200 calorie per day meal replacement diet that included liquid shakes, meal bars, and prepared entrees designed to facilitate a large initial weight loss.
The patients then switched to a diet of conventional foods (of their choosing), with a goal of 1200-1800 calories per day. The physical activity goal was 100 minutes/week of walking or other aerobic activity in the first month, building up to 200 minutes/week by month 6.
At week 68, mean body weight decreased from baseline by 16% in the Semaglutide group vs 5.7% in the placebo group. Almost three times the weight loss with everything else being equal.
Eighty-seven percent of the Semaglutide patients lost more than 5% of their intitial body weight — compared to only 48% of the placebo patients. A significant difference.
About 50% of all patients at the outset of the study were considered to be prediabetic. After 68 weeks, only 7% of the Semaglutide group — compared to 26% of the placebo group — remained prediabetic. These results indicate that Semagluten is more effective in producing weight loss — and with no greater side effects — than any current FDA-approved medication on the market.
Nova Nordisk hopes to obtain FDA approval of the medication my mid-2021. Once approved, it will be administered via a 2.4 mg subcutaneous once-weekly injection. It will be recommended as an adjunct to a reduced calorie diet and increased physical activity. Additional studies suggest that monthly counseling should be sufficient to obtain similar weight losses as those seen in this trial.
====================================================
Source:
An online presentation made at the ObesityWeek® 2020 virtual conference. Presented November 5, 2020 by Thomas A. Wadden, Ph.D.
In-Home Rapid COVID Test Is On The Way
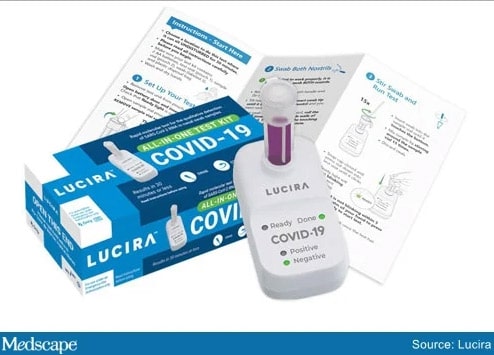
The FDA has recently cleared a COVID-19 test for use in the home. The All-In-One Test Kit is made by Lucira Health and uses single-use nasal swab samples. While the test is also authorized for use in doctor’s offices, hospitals and urgent care centers, the Lucira test can be administered at home to patients over the age of 14. A healthcare professional is required to administer the test to children. It is currently available by prescription only.
“This new testing option is an important diagnostic advancement to address the pandemic and reduce the public burden of disease transmission. It underscores the FDA’s ongoing commitment to expand access to COVID-19 testing,” said FDA Commissioner Dr. Stephen M. Hahn.
The kit includes a test device, a sample vial, a swab, and simple instructions. The device requires two AA batteries. The user is instructed to take the swab and rotate it in each nostril five times. The swab is then stirred in the sample vial. The vial is pressed down onto the test device. Within 30 minutes a positive red or a negative green light flashes.
In tests comparing the Lucira test kit with FDA-approved, high-sensitivity SARS CoV-2 test, the Lucira device matched postive results 94% of the time and negative results 98% of the time. After eliminating tests with very low viral levels, the tests matched 100% of the time.
While Lucira Health scales up its manufacturing capabilities, its COVID-19 test kit will initially be available on a limited basis in healthcare networks that prescribe the test for patients to use at home. The company anticipates that the test will cost around $50.
Does a Facemask Really Protect You?
Yes, you should still wear a mask.

You may have heard of a new study out of Denmark that tested whether paper surgical face masks protect the people who wear them.
The study comes just a week after the CDC updated its guidance on masks to say they aren’t just for the benefit of others, but they also help keep people who wear them from getting sick.
Like a lot of the science on COVID-19, this study has already been passed through the prism of the divided social moment in the U.S., with people interpreting its findings depending on their political leanings.
“Mask wearing doesn’t do a damn thing,” tweeted a conservative talk radio show host in response to the study. But that’s not exactly what the study found. And the bottom line is that you should still wear a mask.
“Masks bring down the community viral load. There’s less getting out, and that means there is less for you to be exposed to,” says John Brooks, MD, a medical epidemiologist in the Division of HIV/AIDS Prevention at the CDC in Atlanta.
In countries where mask use is high, case counts are low. That’s been true throughout history, too. Masks have long been deployed during outbreaks of infectious disease and have been shown to help control the spread of airborne germs.
But Brooks and other experts say the new study is important because it adds to what we know about the population-wide use of masks to control the spread of an airborne disease.
Mask-wearing is considered protective now, but in Denmark, last spring, it wasn’t routine or even advised by public health authorities. In April 2020, researchers split 6,000 Danish citizens into two roughly equal groups. The first group was asked to wear a paper surgical mask anytime they went out in public for the next month.
The study was “powered” ― meaning that it included enough people ― to detect whether following the advice to wear a mask could cut the risk of catching COVID-19 by 50%, or half. It didn’t.
In other words, the trial was designed to look for a big benefit for people who wear masks. It didn’t find that large benefit.
After 1 month, 42 people out of 2,392 in the group that wore masks, or 1.8%, developed a COVID-19 infection, compared to 53 people out of 2,470 in the group that didn’t wear masks, or 2.1%.
That’s a smaller benefit ― about a 16% reduction in infections, on average ― in people who said they wore their masks as they were told. That result didn’t pass a test for statistical significance, though, meaning that it could have been due to chance.
“While it did not reach the 50% threshold that the researchers defined for significance, it’s still a reduction and is consistent with results of other similar studies,” said Linsey Marr, PhD, a professor of civil and environmental engineering at Virginia Tech. Marr is testing the mechanical properties of different kinds of masks and learning exactly how they protect against the spread of the virus. She was not involved with the Danish study.
To get even more specific, the benefits or risks from masks reported in the study ranged anywhere from a 45% reduction in infection for mask wearers to a 20% increase in the risk of getting sick.
There are plenty of reasons not to abandon masks based on this one study.
First, while the study asked people to wear a mask, it didn’t monitor them to make sure that they actually did, or that they wore their masks properly. The researchers tried to account for this by asking people how well they thought they did with compliance. More than half admitted that they didn’t wear their masks perfectly.
“If you walk around any city in the U.S., some people are wearing masks over their noses and other ones are wearing them hanging off one ear,” she says, and that makes the study more of a practical real-world test.
The other important point about the study is that it didn’t test masks as a means of source control, or a way to keep people who are infected from passing the virus on to others.
“If you think about the history, you know the surgical mask was invented not to protect the surgeon from the patient, but to protect the patient from the surgeon,” Marr points out.
In that way, she says, masks are still a really important tool for community protection. “The more people who wear one, the more we’re all protected.”
Marr says the CDC will not change its recommendations based on this research.
=============================================================
Sources:
Henning Bundgaard, PhD, cardiologist, Copenhagen University Hospital, Copenhagen, Denmark.
F. Perry Wilson, MD, associate professor, Yale University; director, Clinical and Translational Research Accelerator, New Haven, CT.
Linsey Marr, PhD, professor of civil and environmental engineering, Virginia Polytechnic Institute and State University, Blacksburg, VA.
Christine Laine, MD, editor-in-chief, Annals of Internal Medicine; clinical associate professor, internal medicine, Jefferson University Hospitals, Philadelphia.
John Brooks, MD, medical epidemiologist, Division of HIV/AIDS Prevention, CDC, Atlanta.
Annals of Internal Medicine: “Effectiveness of Adding a Mask Recommendation to Other Public Health Measures to Prevent SARS-CoV-2 Infection in Danish Mask Wearers.”
Does REM Sleep Help Us Live Longer?

Sleep disturbances effects between 50 to 70 million Americans. Scientists recently took a look at the connection between REM sleep and mortality. REM stands for Rapid Eye Movement and REM Sleep occurs at intervals during the night and is characterized by rapid eye movements, more dreaming and bodily movement, and faster pulse and breathing.
A new study has revealed that less REM sleep is associated with an increased risk for death in middle-aged and older adults. The study, conducted over a 12 year period at the University of California in San Diego, found that each 5% reduction in REM sleep resulted in a 13% increase in mortality rate.
The researchers are quick to point out that such an association does not scientifically establish cause and effect. Both the reduced REM sleep and the higher mortality could be caused by some independent third factor.
“Determining causality can be difficult,” said study investigator Sonia Ancoli-Israel, PhD, professor emeritus of psychiatry at the University of California, San Diego.
Problems with sleep have many known health consequences such as cardiovasular disease; metabolic, psychiatric, and cognitive disorders; and lower quality of life.
The investigation looked at two populations. The first consisted of 2675 men with an average age of 76 years. This group was tracked over a 12 year period. The second group consisted of 1386 individuals, half men and half women, with an average age of 51. They were tracked for 20 years.
All participants in both groups underwent polysomnography and evaluation with the Epworth Sleepiness Scale. Investigators calculated the total number of minutes per night spent in REM sleep and the corresponding percentage of total sleep time. The investigators contacted participants every 4 months to determine their status. When a participant died, the cause of death was categorized as cardiovascular, cancer, and other. Approximately half of the participants died during the course of the study.
What did they discover? The results were dramatic. For each mortality category, the highest percentage of deaths occurred among those with the lowest percentage of REM sleep. For all mortality categories, the mortality rate was higher for participants who had less than 15% REM sleep per night in comparison with individuals who had 15% or more.
Again, this linkage does not necessary indicate that lowered levels of REM sleep cause an earlier death. Overall, the mean percentage of REM sleep was 19.2%. Participants who demonstrated less than this level generally were older; had higher rates of antidepressant use, hypertension, heart attack, and transient ischemic attack; and had less physical activity.
Gender also played a role in the results. When the data were separated by sex, the association between decreased REM sleep and mortality was significant in women but not as much in men.
The researchers pointed out the benefits of promoting sleep health, such as encouraging regular and sufficient nightly sleep, guidance to avoid alcohol before bedtime, and other healthy sleep practices and treating sleep disorders.
The study also revealed the following: The percentage of a person’s REM sleep may be an important, easily interpreted biomarker of aging. Methods are now being developed to allow for the accurate measurement of the percentage of REM sleep using consumer wearable devices, like FitBit and Apple Watch, which make it easier to assess a person’s REM sleep.
However, the investigators caution that although reduced REM does not necessarily predict shorter survival, patients observing this on their fitness device should report this fact to their doctors.
In addition, doctors should regularly ask patients about their quality and quantity of sleep. A sleep study showing reduced time in REM should alert the doctor to consider possible underlying reasons and risk factors for overall poor health.
Interventions to improve sleep quality include promoting regular and sufficient nightly sleep, avoiding alcohol before bedtime, and treatment of known sleep disorders.
If you are suffering from any sort of sleep disorder, be sure to let Dr. Weisz know.
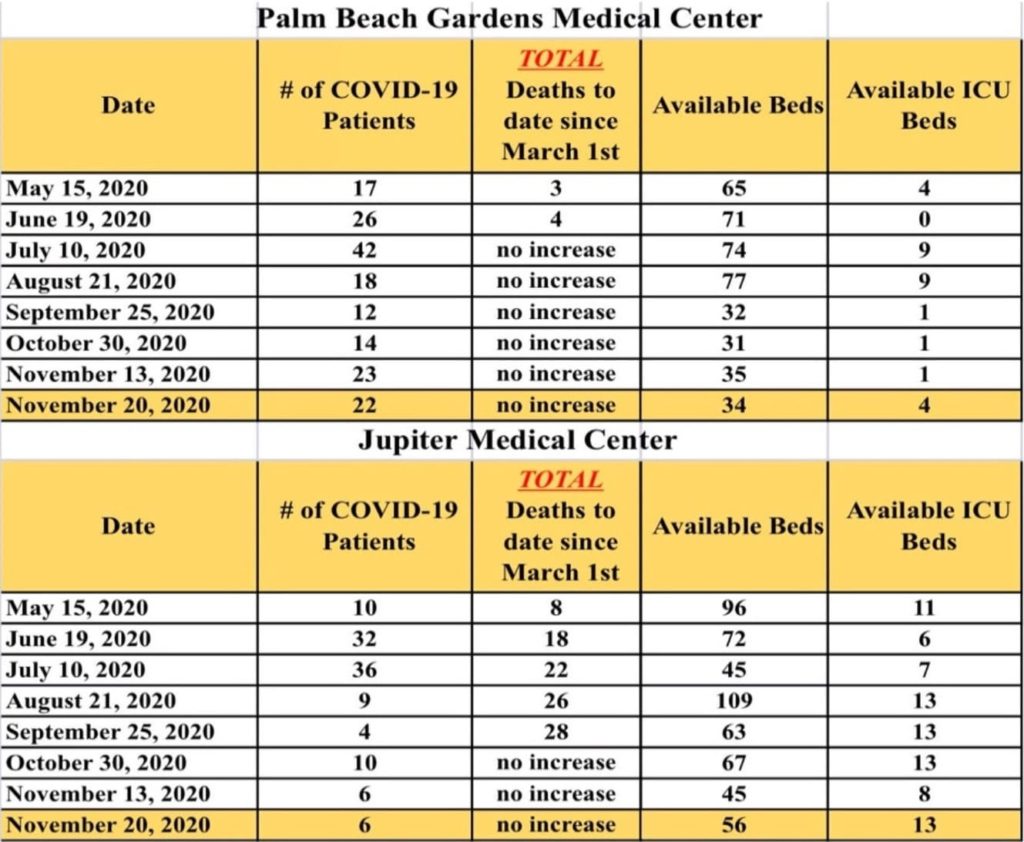
COViD-19 LOCAL SITUATION SUMMARY
Here is the latest update from area hospitals as of November 20, 2020:
LINKS TO LEARN MORE ABOUT CORONAVIRUS:
CDC Corona Virus Prevention Page
Foods That Cause Inflammation Can Lead to Heart Disease

Three new studies have demonstrated that people whose diets include foods with high inflammatory potential are at greater risk of cardiovascular disease (CVD) and stroke.
Individuals who had higher intakes of green leafy vegetables (kale, spinach, arugula), dark yellow vegetables (pumpkin, yellow peppers, carrots), whole grains, fruits, tea, coffee and wine had lower long-term CVD risk than those with higher intakes of red meat, processed meat, organ meat, refined carbohydrates, and sweetened beverages.
After adjustment for use of anti-inflammatory medications, those whose dietary pattern ranked in the highest segment of inflammatory potential had a 38% higher risk of CVD, a 46% higher risk of coronary heart disease, and a 28% higher risk of stroke.
The associations were consistent in all the studies and between sexes and remained significant when adjusted for alcohol consumption, smoking, use of lipid-lowering medications, sodium intake, and blood pressure. This consistency indicates not just an association but also a causality between diet and CVD according to the leader of one of the studies, Jun Li, MD, PhD, at Harvard T.H. Chan School of Public Health and Harvard Medical School in Boston.
“We wanted to be able to provide guidance on dietary patterns and food combinations,” said Li. “If you tell people to eat more polyunsaturated fats instead of saturated fat or trans fat, most people don’t know what foods are higher and lower in those nutrients. Also, many foods have different nutrients — some of which are good and some of which are bad — so we wanted to help people find the foods with the higher proportion of healthy nutrients rather than point out specific nutrients to avoid.”
Dr. Li pointed to extra-virgin olive oil, fatty fish, and tomatoes as examples of foods with established anti-inflammatory activity.
“These studies confirm that an anti-inflammatory diet is useful to prevent cardiovascular events and, more important, that dietary patterns may be made even healthier if subjects increase consumption of foods with the highest anti-inflammatory potential,” she added.
“Mechanistic explanations add plausibility to the results of observational studies.” said Ramon Estruch, MD, PhD, from the Hospital Clinic in Barcelona, Spain. He and his colleagues have suggested that it might be time for better dietary guidelines that take inflammatory foods into consideration.
===============================================================
SOURCE:
J Am Coll Cardiol. Published online November 3, 2020
COVID-19 Medical Update Summary
Pfizer Files for FDA Emergency Use Authorization of COVID Vaccine
November 20:
- Vaccine must be shipped and stored at -70°C (-94°F). Presents logistical storage issues.
- Pfizer and BioNTech seeking approval for vaccines with regulatory agencies around the world.
Operation Warp Speed scientists warn to temper excitement about COVID-19 vaccine’s ability to halt pandemic.
November 21:
- May take year or more to get studies to answer transmission question.
- Studies to date aren’t did not assess transmission. No information on transmission after vaccination at this point.
FDA Authorizes Baricitinib Combo for COVID-19